When you pick up a prescription at the pharmacy, you might be handed a generic pill instead of the brand-name version you asked for. It’s cheaper, sure - but is it the same? That’s where bioavailability and bioequivalence come in. These aren’t just fancy drug industry terms. They’re the scientific backbone that makes generic drugs safe and effective. If you’ve ever wondered why some people swear their generic medication doesn’t work like the brand, or why regulators approve generics without retesting them in thousands of patients, this is the answer.
What Bioavailability Really Means
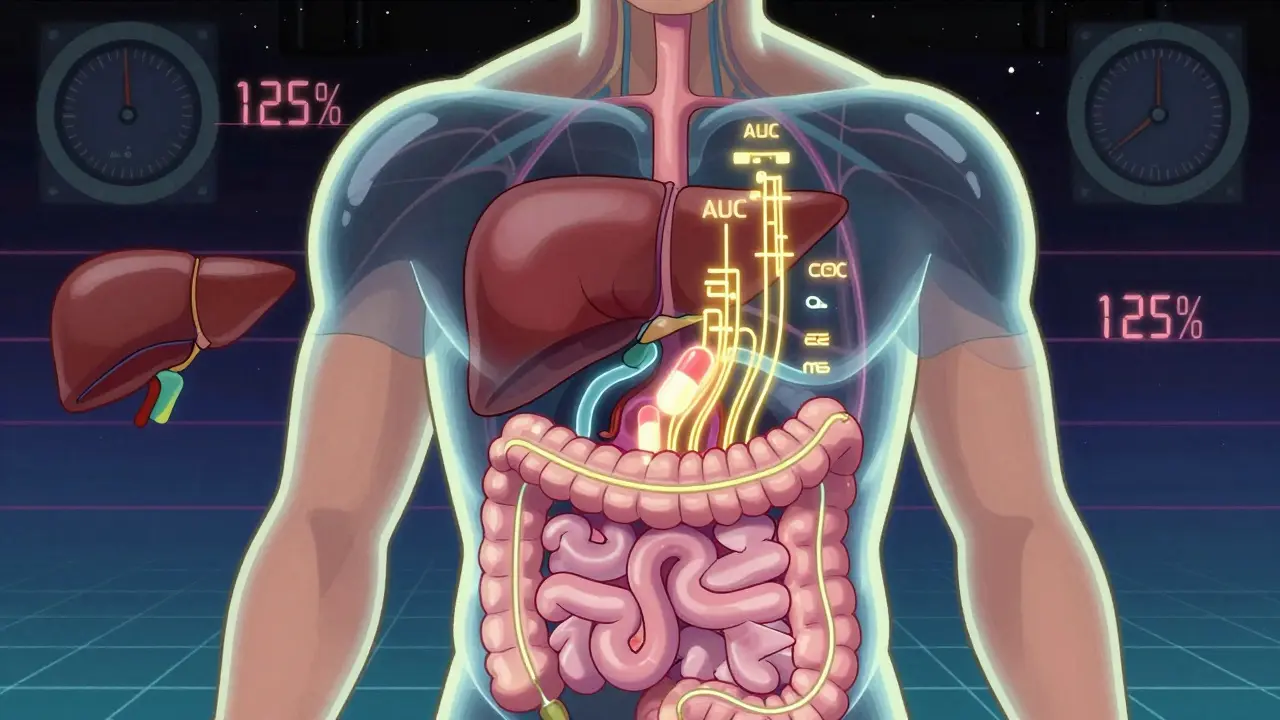
Bioavailability answers one simple question: How much of the drug actually gets into your bloodstream?
Take a pill. It goes through your stomach, gets absorbed in your intestines, then travels to your liver before finally circulating through your body. Along the way, some of it breaks down. Some gets stuck. Some never leaves your gut. The portion that makes it through is your drug’s bioavailability.
For example, if a drug has 60% bioavailability, that means only 60% of the 50 mg you swallowed ends up active in your blood. The rest? Gone. That’s why some drugs need higher doses - not because they’re weak, but because your body doesn’t absorb them well.
There are two types:
- Absolute bioavailability compares the drug’s absorption when taken orally versus given directly into a vein (IV). IV is always 100% because it skips digestion.
- Relative bioavailability compares two different versions of the same drug - say, a brand-name tablet versus a generic one. This is where bioequivalence starts.
The key measurements? Two numbers: AUC (area under the curve) and Cmax (peak concentration). AUC tells you how much drug your body was exposed to over time. Cmax tells you how high the concentration spiked. Together, they paint a full picture of how the drug behaves inside you.
What Bioequivalence Actually Measures
Bioequivalence isn’t about one drug. It’s about two.
When a company wants to make a generic version of a brand-name drug, they can’t just copy the recipe and call it done. They have to prove their version performs the same way in your body. That’s bioequivalence.
Here’s how it works:
- A small group of healthy volunteers (usually 24-36 people) take both the brand-name drug and the generic, in random order, under strict conditions - fasting, same time of day, same water intake.
- Blood samples are taken every 30 minutes to two hours over 72 hours.
- The AUC and Cmax values from both drugs are calculated and compared.
The magic number? 80% to 125%.
That means the ratio of the generic’s AUC and Cmax to the brand’s must fall between 0.80 and 1.25 after statistical analysis. If it does, regulators say the two are bioequivalent. No more testing needed. No need to run trials on thousands of patients with heart disease or diabetes.
This is called the 80/125 rule. It’s not arbitrary. It’s based on decades of data showing that differences smaller than 20% in exposure rarely affect how well a drug works or how safe it is.
Why not 100%? Because drugs aren’t machines. Even two batches of the same brand drug can vary slightly. Your body absorbs them differently on different days. The 80/125 range accounts for normal biological variation.
Why Bioavailability and Bioequivalence Are Different
People mix these up all the time. Here’s the clearest way to tell them apart:
- Bioavailability is a single product’s absorption profile. It’s like measuring how fast your car gets from point A to point B.
- Bioequivalence is a comparison between two products. It’s like saying your car and your neighbor’s car get from A to B in the same time and distance - even if they’re different models.
Bioavailability tells you how a drug behaves. Bioequivalence tells you whether two versions of that drug behave the same way.
Think of it this way: A drug might have 55% bioavailability. That’s fine. But if a generic version has 54% bioavailability - and the 90% confidence interval of the ratio falls within 80-125% - then it’s bioequivalent. The difference is tiny. It doesn’t matter.

When the Rules Get Tighter
Most drugs? The 80/125 rule works perfectly.
But for some, even a 10% difference can be dangerous.
Drugs with a narrow therapeutic index (NTI) are the problem. These are medications where the dose that works is almost the same as the dose that harms you. Think warfarin (blood thinner), levothyroxine (thyroid hormone), or phenytoin (seizure control).
For these, the FDA doesn’t accept 80-125%. It requires tighter limits: 90-111% for AUC in some cases. That means the generic can’t be more than 10% stronger or weaker than the brand.
And it’s not just about numbers. Some drugs behave differently when taken with food. Voriconazole, for example, absorbs 36% more Cmax when taken with a high-fat meal. So the bioequivalence study must include both fasting and fed conditions.
That’s why you’ll sometimes see a generic labeled as “not substitutable” for certain NTI drugs - not because it’s bad, but because regulators demand extra proof.
What the Data Says About Real-World Use
Do generics work? The numbers say yes.
In a 2023 review of over 1,200 patients switched from brand to generic blood pressure meds, only 17 reported problems. Of those, only 4 had confirmed therapeutic issues - and even those were linked to inconsistent dosing, not bioequivalence failure.
Another study of 1,245 patients found that 87.4% noticed no difference at all when switching to generics. Only 3.8% of those who reported issues had their symptoms confirmed as related to the drug - not anxiety, not placebo, not poor adherence.
Still, complaints exist. On forums like Reddit, people report feeling “off” on generic levothyroxine. Some pharmacists say they’ve seen it too. The truth? For NTI drugs, the 80/125 rule might not catch every subtle variation. That’s why some doctors still prefer to keep patients on the same brand - not because generics are unsafe, but because consistency matters more for these drugs.
The FDA and Health Canada both track adverse events linked to generics. Since 2010, fewer than 0.01% of generic approvals have been linked to confirmed therapeutic failures. That’s 1 in 10,000.
How the System Works Behind the Scenes
Generating bioequivalence data isn’t cheap. A single study costs between $150,000 and $500,000. That’s why generic manufacturers often partner with contract labs that specialize in pharmacokinetic testing.
The process is tightly regulated:
- Studies must follow FDA or EMA guidelines.
- Participants are screened for health, weight, and no recent drug use.
- Drug samples are analyzed using high-precision mass spectrometry.
- Statistical analysis is done using log-transformed data because drug absorption doesn’t follow a straight line - it follows a curve.
And it’s not just pills. Complex drugs - like inhalers, topical creams, or injectables - require entirely different testing methods. For example, a generic asthma inhaler might need to prove it delivers the same particle size and lung deposition, not just blood levels.
That’s why some generics still cost more. Not because they’re branded. But because they’re hard to make.
What’s Next for Bioequivalence?
The system isn’t perfect - and it’s changing.
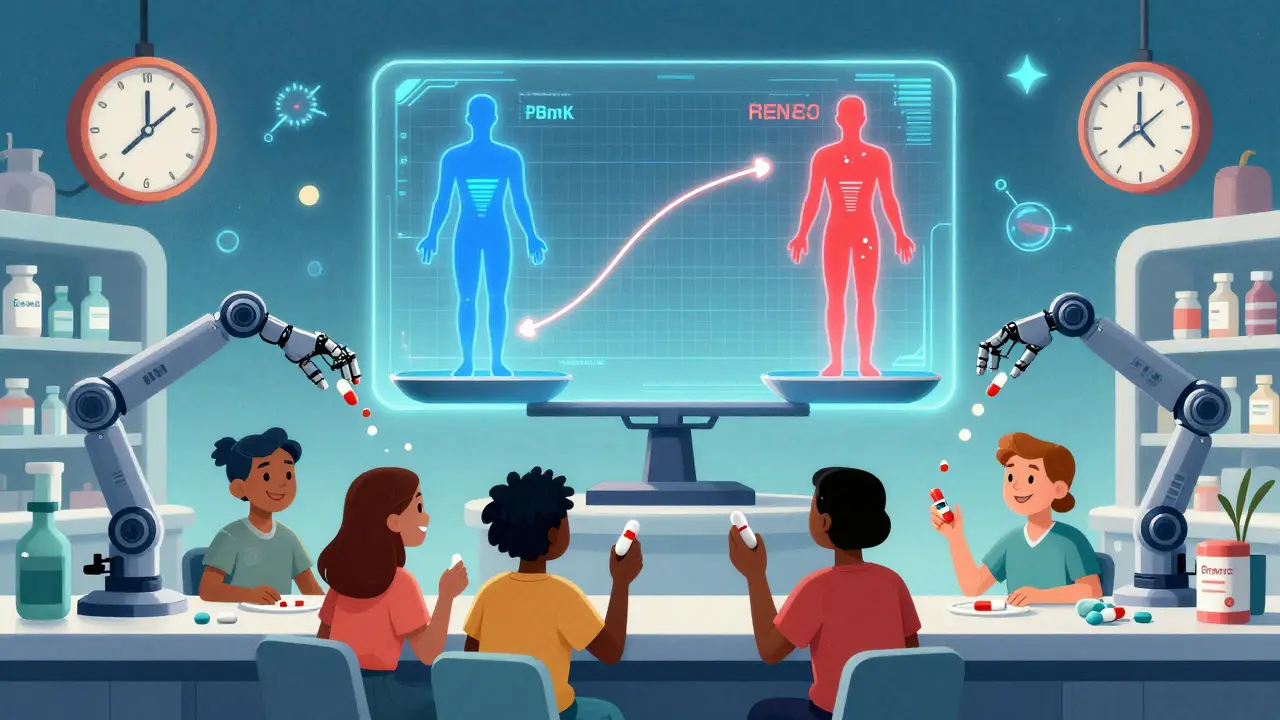
By 2027, experts predict that 30% of bioequivalence assessments for complex drugs will use physiologically-based pharmacokinetic (PBPK) modeling. Instead of testing in 30 people, companies will simulate how the drug behaves in a digital model of the human body. It’s faster. Cheaper. And increasingly accurate.
The European Union is also testing whether in vitro dissolution testing - basically, dissolving the pill in a lab beaker - can replace some human studies. If a generic dissolves the same way as the brand under controlled conditions, maybe we don’t need to draw blood from volunteers every time.
For now? The 80/125 rule stands. And it’s working.
More than 90% of prescriptions in the U.S. are filled with generics. They save patients and insurers billions. And the science behind them? It’s solid.
Are generic drugs really as effective as brand-name drugs?
Yes - if they’ve passed bioequivalence testing. Regulatory agencies like the FDA require generics to deliver the same amount of active ingredient at the same rate as the brand. Studies show 99.7% of generics approved between 2010 and 2020 met this standard. For most people, there’s no noticeable difference in how the drug works.
Why do some people say their generic medication doesn’t work the same?
Sometimes, it’s not the drug - it’s the filler. Generics can use different inactive ingredients, which might affect how fast the pill breaks down in your stomach. For most drugs, this doesn’t matter. But for narrow therapeutic index drugs like levothyroxine or warfarin, even small changes can cause symptoms. If you notice a difference after switching, talk to your doctor. They may recommend staying on the same brand or switching to a different generic.
Is bioequivalence testing the same everywhere in the world?
Mostly yes. The U.S. FDA, European Medicines Agency (EMA), and Health Canada all use the 80-125% range for bioequivalence. The International Council for Harmonisation (ICH) has helped standardize these rules across over 60 countries. Some countries may have minor differences in study design, but the core standards are aligned.
Can I trust a generic drug that costs way less than the brand?
Absolutely. The cost difference comes from not having to repeat expensive clinical trials. The active ingredient, dosage, and absorption profile are legally required to match the brand. A $5 generic isn’t a low-quality version - it’s a scientifically approved alternative. The savings come from competition, not cutting corners.
What should I do if I think my generic drug isn’t working?
Don’t stop taking it. Talk to your doctor or pharmacist. Keep a log of symptoms, timing, and when you switched medications. Sometimes, the issue is unrelated - like stress, diet, or another condition. If your doctor suspects a bioequivalence issue, they can request a blood test to check drug levels or switch you to a different generic. For NTI drugs, staying on the same manufacturer is often the safest approach.
John Smith
So let me get this straight - you're telling me I can swap my $200 brand-name pill for a $5 generic and my body won't even notice? That's wild. I've been on levothyroxine for 8 years and my endo still makes me stick with the brand. Guess I'm just one of those 3.8% who 'felt off' - or maybe the 0.01% where the math didn't quite hold up. Either way, I'm not risking my thyroid on a gamble.
Also, why does the FDA let generics use different fillers? That's like saying two cars have the same engine but one has a plastic gas tank and the other has steel. One might work fine until it doesn't.
Sharon Lammas
I think what's missing here is the emotional weight behind this. People aren't just talking about pharmacokinetics - they're talking about trust. After years of relying on a medication that kept them alive or stable, switching to something that looks different, tastes different, even feels different... it's not irrational to feel uneasy. The science says it's equivalent. But the body remembers. And sometimes, that memory matters more than the data.
Donna Zurick
I work in a pharmacy and I've seen this firsthand. Patients panic when they get a different-looking pill. We hand them the same drug, just from a different manufacturer, and they ask if it's real. We need better education. Not more studies. Just clearer info. Also, side note: the generic for my blood pressure med tastes like chalk. That's not bioequivalence, that's a sensory betrayal.
Pankaj Gupta
The 80-125% rule is statistically sound, but it assumes homogeneity in human physiology. In reality, gut microbiomes vary wildly across populations. A drug that behaves predictably in a cohort of 30 healthy American volunteers may behave differently in someone with chronic inflammation, a fungal overgrowth, or a diet high in turmeric. The system works well for the majority, but fails the outliers. And outliers still deserve care.
Richard Elric5111
It is an undeniable fact that the regulatory framework governing bioequivalence is predicated upon a reductionist paradigm that prioritizes quantitative metrics over qualitative lived experience. The reliance upon AUC and Cmax as primary endpoints constitutes a epistemological limitation, inasmuch as it neglects the phenomenological dimension of pharmacological response. One may achieve statistical equivalence without achieving therapeutic congruence.
Dean Jones
I read the whole thing twice. Let me break it down. Bioavailability is how much of the drug gets into your blood. Bioequivalence is whether the generic gets in about the same amount as the brand. The FDA says if it's within 20% either way, it's good enough. But here's the thing - 20% isn't nothing. For a drug where 5mg is the sweet spot and 6mg makes you dizzy, that 20% is the difference between feeling normal and feeling like you're on a rollercoaster. And nobody tells you which generics are made by which company. I switched from Mylan to Teva and suddenly I was dizzy every morning. My doctor said, 'It's bioequivalent.' Yeah, but my body didn't get the memo.
Betsy Silverman
I used to work in clinical trials. We did bioequivalence studies. The volunteers were paid, fasting, monitored 24/7. Real world? People take pills with coffee, with food, at 2am, forget to take them for two days, then double up. The lab conditions are perfect. Life is not. That’s why some people notice a difference. It’s not the drug. It’s the context. And we act like it doesn’t matter.
Ivan Viktor
So let me get this straight. We test a drug on 30 people in a lab, then approve it for 30 million. And we call that science? I've seen a generic version of my antidepressant that looked like a child's drawing of a pill. It worked. Then it didn't. Then it did again. I'm not a scientist. I'm just a guy trying not to cry in the shower. Can we maybe just... admit that biology is messy?
Zacharia Reda
You know what's wild? The companies that make generics are the same ones that make the brand drugs. They just slap a different label on it. So when you say 'the brand works better,' you're really saying 'I prefer the version with the blue pill and the fancy packaging.' The science doesn't care. Your brain does.
Jeff Card
I had a cousin on warfarin. Switched to generic. INR went from 2.4 to 4.1 in two weeks. Almost bled out. They said it was 'within range.' But 4.1 isn't 'within range' if you're supposed to be at 2.5. They didn't test for that. They just tested AUC and Cmax. But warfarin doesn't care about those numbers. It cares about clotting time. So yeah. Sometimes the rules miss the point.
Matt Alexander
Simple version: brand = original recipe. generic = copycat. If the copycat tastes the same and gives you the same buzz, it's fine. If not, try a different copycat. Not all generics are made equal. Some use better fillers. Some use cheaper ones. It's like buying a Ford vs. a Chevy. Same car, different parts inside.
Gretchen Rivas
For NTI drugs, consistency matters more than equivalence. If your thyroid med works, stay on it. Don't switch unless you have to. And if you do, ask for the same manufacturer. That's the real secret the FDA won't tell you.
Stephen Vassilev
I've been researching this for 14 years. The truth is, the FDA doesn't test generics at all. They rely on data submitted by manufacturers who have financial incentives to manipulate dissolution profiles. There are whistleblower reports. There are internal memos. There are documents buried in FOIA requests that show how some generics failed dissolution tests... and were approved anyway. And now, with PBPK modeling on the horizon? We're moving toward a system where algorithms decide if a pill is safe... based on simulations built by the same companies that profit from its sale. This isn't science. It's a carefully engineered illusion.





Write a comment