
The U.S. Food and Drug Administration doesn’t call ahead. No warning. No schedule. Just a knock on the door - sometimes early in the morning, sometimes during shift change - and suddenly, FDA inspectors are walking through your facility. This isn’t a drill. It’s standard procedure. And if you’re making medicine, medical devices, or food products sold in the U.S., you need to be ready.
Why the FDA Shows Up Without Warning
The FDA doesn’t conduct unannounced inspections just to catch companies off guard. They do it because announced inspections don’t tell the truth. When a facility knows inspectors are coming, they clean up. They fix the obvious issues. They pull out the best records. But real compliance? That’s what happens on a Tuesday at 3 p.m. when no one’s watching.
Before May 2025, the FDA mostly inspected domestic facilities without notice. Foreign facilities? They got weeks of warning. That created a dangerous double standard. A factory in China might have had time to hire extra staff, rewrite logs, or hide defective batches. Meanwhile, a plant in Ohio had to operate under full scrutiny every time. The FDA found serious problems in foreign facilities more than twice as often when they had advance notice.
On May 6, 2025, the FDA changed that. They expanded unannounced inspections to cover all foreign manufacturing sites supplying products to the U.S. - no more heads-up. Why? Because 40% of finished drugs and 80% of active pharmaceutical ingredients used in America come from overseas. If you’re not inspecting those facilities the same way you inspect U.S. ones, you’re leaving Americans at risk.
What Happens When They Walk In
When an FDA investigator arrives, they show ID. They hand you Form FDA 482 - the official Notice of Inspection. They don’t ask for permission. They don’t wait for the CEO. They talk to whoever’s in charge at that moment - shift supervisor, quality lead, even the night janitor if they’re the only one around.
Then they start looking. Not at the shiny new equipment. Not at the cleaned-up production line. They dig into:
- Standard Operating Procedures (SOPs) - are they followed, or just printed and hung on a wall?
- Manufacturing records - are batch logs accurate, or were they backfilled after the fact?
- Training records - did that technician really get trained on sterile handling, or did they just watch a 5-minute video?
- Complaint handling - are customer complaints logged and investigated, or buried?
They might ask to enter a sterile manufacturing area. They might request raw data from a chromatography system. They might compare your internal audit reports to what’s actually happening on the floor. If you’re using outdated software, paper logs, or inconsistent procedures - they’ll find it.
Inspections can last hours or days. It depends on the size of the facility, the complexity of the processes, and what they find. Even if they issue a “No Action Indicated” report, that still matters. It tells the FDA your facility is trustworthy. And trust matters when you’re competing for contracts.

Why Foreign Facilities Got a Free Pass - Until Now
For years, the FDA coordinated foreign inspections with local health authorities. They’d send a heads-up to India’s drug regulator. They’d schedule visits around holidays. They’d even invite foreign inspectors to tag along. That made logistics easier - translators, travel, paperwork. But it also made inspections predictable.
Companies in India, China, and elsewhere began using that lead time to their advantage. They’d hire temporary staff. They’d fix broken equipment. They’d scrub digital records. One manufacturer told a trade publication they had “two weeks to make everything look perfect.” That’s not compliance. That’s theater.
The FDA’s own data showed that when foreign facilities were given advance notice, inspectors found serious violations in over 90% of inspections. Domestic facilities? Only about 45%. That gap wasn’t about skill - it was about opportunity. And the FDA decided to close it.
What Manufacturers Need to Do - Now
You can’t prepare for an unannounced inspection. But you can be ready for it. Here’s what works:
- Build compliance into your daily routine - not just before audits. If your quality management system (QMS) is only updated when someone’s coming, you’re already behind.
- Use digital tools with audit trails - paper logs can be altered. Digital systems that track who changed what, when, and why? Those are harder to fake.
- Train everyone - not just QA - operators, maintenance, warehouse staff. If an inspector asks a line worker why a batch was rejected, they should know the answer.
- Run mock inspections weekly - simulate surprise visits. Turn off the lights. Send someone in unannounced. See what breaks.
- Know your records cold - if you can’t pull a batch record in under five minutes, you’re not ready.
The FDA doesn’t expect perfection. But they do expect honesty. If your SOP says you clean equipment after each run - then clean it. If your training record says someone completed a course - make sure they did. Falsifying records? That’s a red flag. And the FDA’s new policy is laser-focused on catching those who try to hide violations.
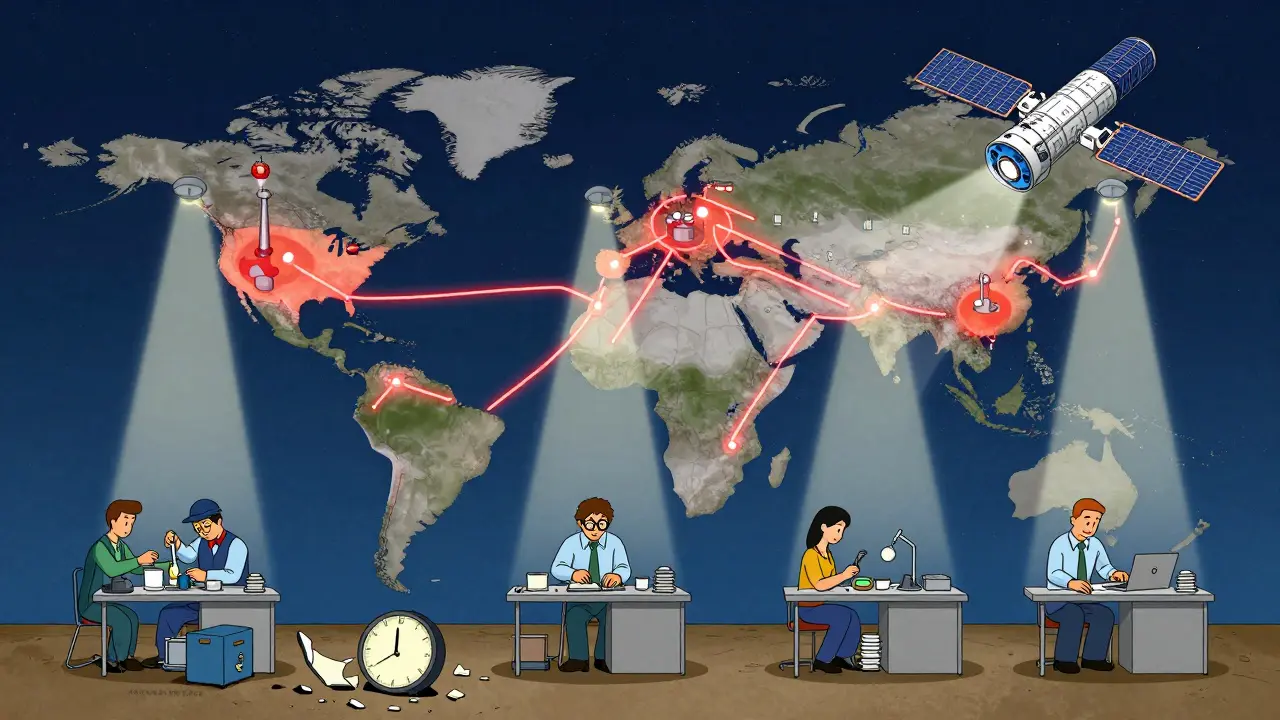
The Bigger Picture: Global Supply Chains and Public Health
This isn’t just about rules. It’s about safety. Think about the last time you took a prescription drug. Did you know its active ingredient might have been made in a factory 8,000 miles away? Or that your IV bag was filled in a facility that never had a surprise visit until last year?
The FDA’s move to unannounced inspections abroad is a direct response to globalization. As supply chains stretch across continents, oversight must tighten. It’s not about punishing foreign manufacturers. It’s about leveling the playing field. Every facility - whether in Kansas or Karnataka - should be held to the same standard.
President Trump’s May 5, 2025 executive order called for stronger oversight of foreign drug makers. The FDA’s policy change is the enforcement arm of that order. And it’s working. Early reports show inspections in India and China are now happening with zero notice - and findings are more consistent with U.S. results.
But challenges remain. Language barriers. Cultural differences. Coordination with foreign regulators. The FDA hasn’t said yet whether they’ll still notify local authorities before showing up. That’s still being worked out. But one thing is clear: the days of advance notice for foreign facilities are over.
What Comes Next
The FDA says this is a permanent shift. Not a pilot. Not a test. A new era in enforcement. They plan to increase the frequency of unannounced inspections globally. They’re hiring more investigators. They’re training them in international protocols. And they’re not backing down.
For manufacturers, this means one thing: compliance can’t be seasonal. You can’t turn it on when you’re shipping a new product. You have to live it every day. Because the FDA isn’t coming to inspect your paperwork. They’re coming to inspect your reality.
If your facility is ready for a surprise visit today - you’re not just compliant. You’re protected. And in a world where one contaminated batch can shut down a global supply chain, that’s not just smart. It’s essential.
Are all FDA inspections unannounced?
No. Most inspections are unannounced, but there are exceptions. For example, the FDA may give advance notice for inspections related to certain medical device classes (like Class II or III devices) to ensure key personnel are available. They also sometimes notify facilities for follow-up inspections or if language or logistical barriers require coordination. But for routine quality system audits - especially for drugs and food - surprise visits are the norm.
What happens if a facility refuses an unannounced inspection?
Refusing an FDA inspection is a serious violation. The agency has legal authority to block products from entering the U.S., issue warning letters, or even pursue criminal charges. The FDA can also detain shipments, revoke facility registrations, or ban future imports. In practice, most facilities comply because the consequences of refusal are far worse than the findings of an inspection.
Do foreign manufacturers still get help from local regulators during inspections?
It’s unclear. Before May 2025, the FDA often coordinated with foreign regulatory bodies - sharing schedules, inviting observers, and using local translators. Since the shift to unannounced inspections, the FDA hasn’t confirmed whether this practice continues. Some manufacturers report receiving no prior notice from any authority, while others say local agencies still get informed. The lack of clarity is a current point of concern for global manufacturers.
How often do FDA inspectors visit foreign facilities now?
The FDA conducts about 3,000 inspections annually at foreign facilities across more than 90 countries. Since the May 2025 policy change, they’ve increased the frequency of unannounced visits, especially for high-risk products like injectables, sterile devices, and active pharmaceutical ingredients. There’s no fixed schedule - inspections are risk-based, meaning facilities with past violations or complex processes are targeted more often.
Can a company be fined or shut down after an unannounced inspection?
Yes. If inspectors find serious violations - like falsified records, contaminated products, or failure to follow approved processes - they issue a Form 483 listing the issues. If the company doesn’t respond adequately, the FDA may issue a Warning Letter. Continued non-compliance can lead to import bans, product seizures, or even criminal prosecution. Facilities with repeated violations may be placed on import alert, meaning all their products are automatically detained at U.S. ports.
tynece roberts
so like… the FDA just shows up and you better have your shit together or else? yeah i get it but honestly this feels like micromanaging gone wild. we’re talking about people who make life-saving meds here, not some sketchy vape shop. why not trust them to do the right thing instead of playing gotcha? 🤷♀️
Rosemary Chude-Sokei
While I appreciate the intent behind unannounced inspections, I must emphasize that consistency in regulatory standards across jurisdictions is paramount. The global pharmaceutical supply chain operates under complex, multilayered compliance frameworks. Abrupt policy shifts without harmonized international coordination risk creating operational chaos, especially for SMEs in developing economies who lack the resources to maintain redundant compliance infrastructure. We must balance vigilance with equity.
mir yasir
As someone who works in pharma manufacturing in India, I can tell you this policy is long overdue. For years, we were expected to jump through hoops while our competitors abroad got a free pass. The ‘two weeks to make everything look perfect’ mentality was systemic. Now, inspectors are seeing real operations - not performances. That’s not punishment. That’s justice.
Yes, it’s harder. Yes, it costs more. But if your facility can’t operate safely without a warning, then you were never truly compliant. We’ve adapted. Our quality systems are now embedded in daily work - not just audit season.
And let’s be clear: this isn’t about targeting India or China. It’s about protecting patients everywhere. If your insulin comes from a facility that only cleans up when they know someone’s coming - that’s not a national issue. It’s a global health crisis waiting to happen.
Leah Dobbin
How utterly predictable. Of course, the FDA would finally catch on that foreign manufacturers were gaming the system. It’s almost embarrassing how long it took them to realize that ‘globalization’ doesn’t mean lowering standards - it means enforcing them universally. The fact that this was even a debate speaks volumes about the complacency of regulatory bodies over the last decade. Honestly, it’s a miracle we haven’t had a major contamination scandal on this scale.
Hugh Breen
OMG YES. 🙌 This is the kind of bold, no-nonsense leadership we need. 🌍💉 I’ve been saying this for YEARS - if you’re making medicine for Americans, you play by AMERICAN RULES. No exceptions. No ‘but in my country we…’ nonsense. The FDA just dropped the mic. 🎤💥
Let’s be real - this isn’t about politics. It’s about not letting people die because someone thought it was easier to fake a log than fix a machine. I’m so proud of them. Send more inspectors. Hire more people. I’ll fundraise for their coffee.
Byron Boror
Finally. Took long enough. This country’s been getting ripped off for decades. Foreign factories get a free ride while American workers get crushed under compliance costs. Now they’re forced to play fair. Good. Let ‘em sweat. If they can’t meet U.S. standards, they shouldn’t be selling here. Period. No more hand-holding. No more ‘cultural sensitivity.’ Just clean rooms, real records, and zero excuses.
Stephanie Paluch
as someone who’s worked in QA for 12 years… this is the most common thing i see: people think compliance is a checklist. it’s not. it’s a culture. if your janitor doesn’t know why sterile technique matters, you’re one sneeze away from a recall. the FDA’s move isn’t scary - it’s necessary. 🤗
Buddy Nataatmadja
Interesting shift. I’ve been in a few foreign facilities - India, Vietnam, Mexico - and honestly, the quality was often better than expected. But you’re right: the *consistency* was the issue. Some places had world-class labs and zero documentation. Others had perfect paperwork and broken autoclaves.
Unannounced inspections expose that gap. It’s not about distrust - it’s about transparency. If you’re proud of your process, you shouldn’t fear a surprise visit. You should welcome it. Because real quality doesn’t need a spotlight.
Lorna Brown
There’s a philosophical question here we’re avoiding: If compliance is only meaningful when it’s enforced unpredictably, does that mean ethical behavior is contingent on surveillance? Or is this simply the necessary evolution of trust in a globalized, high-stakes system where incentives are misaligned?
Perhaps the real innovation isn’t the inspection model - it’s the recognition that human behavior cannot be regulated by policy alone. You can’t audit integrity. But you can build systems that make dishonesty exponentially harder than honesty.
Rex Regum
Oh please. This is just another government power grab disguised as ‘public safety.’ Who authorized the FDA to start treating foreign manufacturers like criminals? They’re not breaking the law - they’re following THEIR laws. This is cultural imperialism wrapped in lab coats.
And don’t even get me started on Trump’s ‘executive order.’ You think this was about safety? No. It was about nationalism. ‘America First’ means ‘foreigners must bow’ - not ‘let’s make medicine safer.’
Next they’ll be inspecting your kitchen for not using organic salt.
Kelsey Vonk
Reading this made me think about how much of our daily life depends on invisible systems - the water we drink, the pills we take, the vaccines we rely on. We don’t think about them until something goes wrong. And when it does? It’s never just one person’s fault.
Maybe this isn’t about punishment. Maybe it’s about honoring the people who show up every day to make sure things are safe - even when no one’s watching. That’s worth protecting.
Emma Nicolls
so like… i work in a small med device shop and we just got our first unannounced visit last month and honestly? it was kinda chill. they asked questions, looked around, didn’t yell. we had our records ready because we do audits every week. no drama. just… good work. 🤞
if you’re doing the right thing daily, surprise visits aren’t scary. they’re just… confirmation. like a pat on the back from the government. lol
Richard Harris
Interesting development. I’ve worked with EU and US regulators over the years, and while I appreciate the intent, I wonder if the lack of coordination with local authorities might inadvertently create friction. Language barriers, cultural norms around authority, and even time-zone logistics can hinder effective communication. Perhaps a phased approach - with limited prior notification for logistical purposes - might improve compliance without compromising integrity.
Also, typo: ‘pharmaceutical’ - I meant ‘pharmicuetical’… oops.
Kandace Bennett
FINALLY 😍👏 The FDA did what they should’ve done YEARS ago. 🇺🇸💊 You can’t have a ‘global market’ where half the players are cheating. This isn’t about being mean - it’s about being fair. And if you’re making medicine for Americans? You better be as good as the ones in Ohio. No excuses. No ‘but in my country…’ nonsense. 🙅♀️
Also - anyone else think the FDA should start publishing inspection results like Yelp? ‘5 stars: clean facility, great SOPs!’ ‘1 star: fake logs, expired gloves, janitor knew more than QA.’ 😏




Write a comment